Understanding Calories & How They Relate to Weight Loss
The word ‘calorie’ is a ubiquitous part of our dietary vocabulary yet are very much an abstract idea and is hard to grasp how it affects weight loss. You can’t see calories, touch them or sense them but unfortunately you can see their effects all too well if you eat too many of them. As omnipresent as calories are in our lives, and in the minds of those determined to lose weight, they are perhaps the least understood aspect of nutrition for most dieters, which is ironic given the fact that an understanding of calories and their role is crucial to anyone serious about losing weight. The confusion over calories comes mostly thanks to the bombardment of contradictory information from food manufacturers, the weight loss industry and the media. Food manufacturers spend billions in advertising dollars to disseminate material that promotes their products and undermines any negative health aspects surrounding the foods they make. The weight loss industry profits off of products and services that help you lose weight temporarily, as any long term solution would inherently mean no repeat business and eventually dwindling profits, and so they create and support items based on trends that come and go. The media on the other hand, uses dietary information as a form of entertainment since weight loss based material is one of the most popular topics with consumers and uses it to increase audience numbers rather than as a tool for educating the public. Both food manufacturers and the weight loss industry have a vested interest in telling us a lot about nothing. Which leaves most people bewildered by what to do next in terms of their weight loss and frankly begin to distrust the seemingly ever changing messages from what they are lead to believe are science based authorities on weight loss. Having lost faith in science, to paraphrase Chesterton, when people don’t believe in science they do not thereafter believe in nothing, they then become capable of believing in anything. Which is exactly what the food and weight loss industry needs for consumers to buy more of their products and services. In today’s world an educated consumer wouldn’t be much of a consumer since an understanding of the science behind calories would make them quite wary of popular diet and exercise trends. An educated consumer would also never fall for the calorie related strategies used by food corporations that are meant to make you buy their products for the wrong reasons. (Read my article on the How The Food Industry Makes You Overeat- The Economics Of Obesity for more information on how marketing confuses consumers.) Calories aren’t rocket science, but they are a bit complicated. Nevertheless, if you take the time to understand this fundamental element of nutrition, it will help you go a long way in achieving your weight loss goals and being very much a well educated consumer who will make better choices.
Understanding Calories- The Nomenclature

One of the most bewildering problems is the word itself as the word calorie as it appears on food labels isn’t exactly the correct term, even though it’s widely used. There is a saying that you can’t understand the nature of anything unless you call it by its proper name and I think this precept is applicable in this case as well. A calorie isn’t the correct label for the energy in our foods. A calorie (written with a lower case ‘c’) is a measurement of heat energy used in thermodynamics and is never used in regard to food energy measurement. The energy in our foods is measured in kilocalories, (k/cal), which means 1,000 calories with the word ‘Calorie’ (with a capital ‘C’) used by those in the dietary fields to refer to kilocalories. Are you confused yet? A medium sized apple therefore isn’t really 80 calories in the strictest sense, it’s 80 kilocalories or 80,000 calories! It makes sense to keep the numbers manageable by expressing all values in units of a thousand and I am certain that food manufacturers would be horrified to see their products labeled with three extra zeros added to the calorie count. Other countries outside the United States avoid the whole conundrum by using the metric measurement of kilojoules, which is 4.18 kilocalories. From here on in for the sake of simplicity I will use the correct nomenclature- Calorie or kilocalorie when referring to units of food energy measurement so we are all on the same boat.
What Is A Calorie/Kilocalorie?

A kilocalorie is the amount of energy required to increase the temperature of one liter of water by one degree Centigrade from 14.5 degrees to 15.5 degrees Centigrade. A wonderful definition, but one that means little to anyone without a background in chemistry or an understanding of the metric system. To translate for those who didn’t grow up with meters and grams, a Calorie/kilocalorie is the amount of heat needed to raise a quart of water (which is just about a liter) by 33.8 degrees Fahrenheit – which is 1 degree Centigrade. Water boils at 212 degrees Fahrenheit (100 degrees Centigrade). Putting this into perspective, if the energy intake of the average American adult male is 2437 Calories daily [1], that would be enough energy to boil a little over 6 gallons of water! The amount of energy is astounding, but it you must bear in mind that unlike the energy released in a fire, the energy from foods is released very slowly through the processes of digestion and metabolism. If not, we would have some serious problems to contend with and spontaneous combustion would be a regular event! Where does all this energy go? Mostly towards the biological work required for our bodily functions, everything from breathing to using our muscles and repairing our cells. This energy is used to maintain balance (homeostasis- a bigger word with the same meaning) with the heat energy released from these functions serving to maintain our body temperature. (See my article on metabolism here).
Measuring Calories/Kilocalories and How It Relates to Weight Loss

So how do we measure Calories/Kilocalories? The process actually isn’t that complicated as the food is burned to ash and the heat released is measured in a device called a bomb calorimeter. Since the Third Law Of Thermodynamics states that energy can be neither created or destroyed, we know that the amount of energy released from a food inside our body through digestion and oxidation would be about the same as the amount of energy released if it was completely burned in a closed system. A bomb calorimeter does just that and researchers make allowances for the components of food that would not be completely digested in your body. Some plant-based fibers, for example cannot be completely digested and thus can’t be converted into energy. As result any calories from indigestible food components are subtracted from the total amount of heat energy measured. Also subtracted are the nitrogen components of protein-based foods which are also excreted by our bodies without being used. By taking these factors in to consideration researchers can arrive at a final figure that best represents what our body would gain from a food. Without these corrections you would find many low-calorie foods like vegetables being listed as high calorie foods with a much higher Calorie/kilocalorie count than they really do. Thanks to the requirements of the provisions of the Nutrition Labeling and Education Act enacted in 1990, all foods in the U.S. must have their nutritional information listed prominently on the container in which they are sold. These labels are often misleading in terms of their calorie counts as while the kilocalorie values are accurate, the serving sizes are kept ridiculously low so that the calorie count won’t appear to be too high. Take the label on a small bag of a brand of ‘natural’ potato chips for example with its serving size of 1 ounce which has a value of 150 Calories (kcal). Now 150 Calories may not sound like much, but that’s only one seventh of the total bag, and I can’t tell you the last time I saw anyone eat one seventh of a small bag of potato chips as most people would eat the whole bag. Thus, the real energy yield is more like 1050 Calories, which is almost half the average number of kilocalories needed by a woman to maintain her bodyweight and is a glaring example of a seriously high calorie food masquerading as a harmless low calorie treat thanks to serving size manipulation.
The Caloric Components Of Carbohydrates, Proteins and Fats

Now the energy content of the food we eat come from three macronutrients- carbohydrates, protein and fats. Protein and carbohydrates for the most part have a total of 4 kilocalories per gram, (a gram is about one third of an ounce by the way), whereas fats have 9 kilocalories per gram. Consequently, foods that are high in fat are usually high in calories. Olive oil has many heart healthy benefits, but it is still very much a high calorie food. As little as two tablespoons yields 28 grams of fat, which adds up to 248 kcal. In other words, two tablespoons of olive oil are calorically equal to more than three medium sized apples. That’s why you have to be careful about eating foods high in fats. Not because fats are bad but because it’s just so easy to consume a lot of calories by eating just a little of it and the way our brains work, we tend to want more of it. So when choosing foods it’s important to know the fat content and watch your the portion sizes if it is high in fat as the numbers can easily add up quickly to a point where you take in more calories than your body needs- which can make you gain weight.
(The mathematically savvy amongst you may have discerned that while olive oil has a total of 28 grams of fat- it only has 248 kilocalories and not 252 kilocalories. If 1 gram of fat yields 9 kilocalories, then it should have 252 kilocalories- (9X28=252) but it doesn’t. The reason for the discrepancy is that oils (and most foods) contain some amount of water which is calorically inert.) Why do carbohydrates and proteins have only 4 kilocalories per gram, whereas fats have 9 kilocalories per gram? The answer isn’t that hard to fathom if you keep in mind the basic elements and molecular makeup of these nutrients. Protein is made of amino acids while carbohydrates is made up primarily of glucose, both rather simple structures on a molecular level. Our bodies can’t use proteins, carbohydrates and fat as they are, since they must be broken down into their individual elements before being absorbed by the body or used as an energy source. This requires energy and since fats are composed of chains of fatty acids which are far more complex than carbohydrates or proteins in terms of biochemical structure and the number of chemical bonds holding it together, more energy is released in breaking it down compared to the energy released in breaking down protein into amino acids or carbohydrates into glucose. If you conceptualize the idea of energy being released when a food is broken down, you can also understand why fats are such a good energy source as fatty acids which have far more bonds holding it together than glucose or amino acids can thus store more potential energy.
Calories/Kilocalories And Losing Body Fat
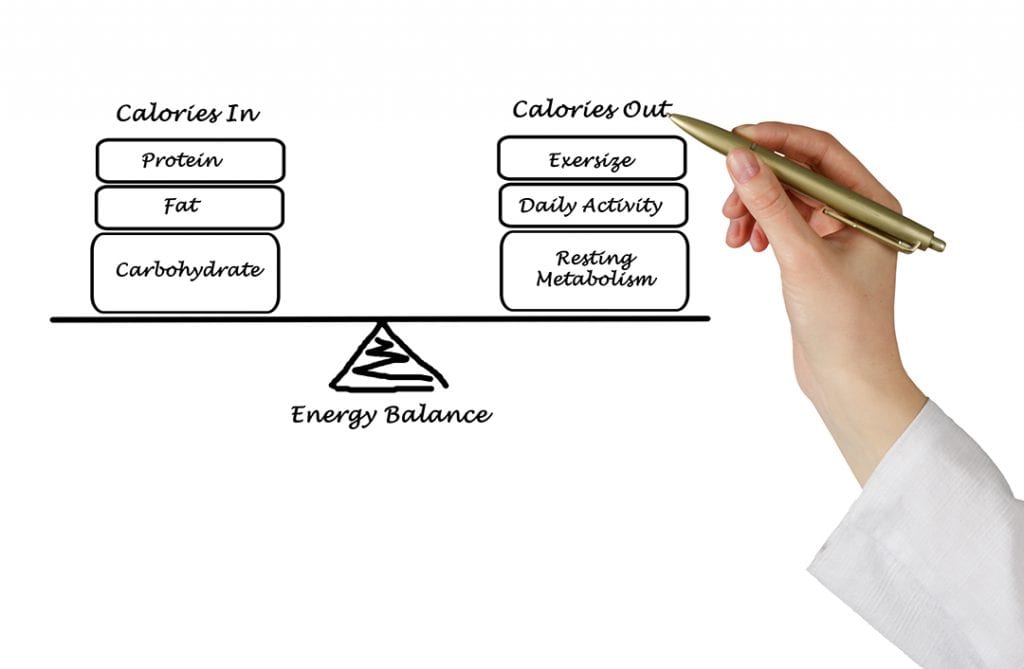
One of the most important themes from the laws of thermodynamics is the idea that if we consume more Calories than we need our bodies will have no choice but to store them as body fat. This will happen regardless of how healthy the food source may have been. You can get fat from eating high protein foods, fats or carbohydrates- even if it is labeled fat free, organic or heart healthy. It makes no difference whatsoever the type of food you eat, if you ingest more than you should, you’ll gain weight. But as we said before, it is easier to go overboard calorie-wise from eating foods that are high in fat since they are so much more calorically dense. I should mention as well that at 7 kcal per gram, alcohol has more calories than protein and carbohydrates and the added curse of being processed in such a way that if you consume too much of it can significantly increase fat stores around your liver and other organs. (See my article on alcohol and weight loss ).

Why You Can’t Lose 10 Lbs. Of Fat In A Week
One of the best aspects of understanding what calories are and how it applies to our daily lives is that it gives you an informed perspective as to how ludicrously unrealistic rapid weight loss plans can be. The Holy Grail of losing 10 pounds of fat in a week is highly improbable, given the very concrete thermodynamic laws involved and even losing 10 pounds of fat in a month is still outside the realm of reasonable expectations. Consider that a pound of fat contains approximately 3,500 Calories (kilocalories), which is a quite a lot of stored energy when you put it in perspective. The average male needs anywhere in the ballpark of 2,000 to 3,000 Calories a day to maintain a stable body weight, a figure that is slightly lower for females, but it gives you an idea of just how much 3,500 Calories really are. As non obese men drinking nothing but water can lose no more than 4 kg of bodyweight (almost 9 lbs) over a 7-10 day period. [6,7] Keep in mind that this is just bodyweight and of that 4 kg most of it is fluid and the remainder being some lean muscle tissue and some fat stores. Thermodynamically you might think that increasing activity through exercise while starving will do it, but that will only make you end up in an emergency room or worse, as it is physically impossible to do the type of strenuous activity without food intake. And keep in mind that after such extreme measures, the loss of muscle mass will be such that overall metabolic rate will be lowered, which means that you would end up having to eat less afterwards to avoid being fatter than you were before such an extreme fast. Fat loss is a gradual phenomenon and can’t be rushed as it simply a matter keeping your caloric intake within an acceptable level while increasing your body’s need for more energy. You can do this by exercising and increasing your muscle mass, since muscle requires additional calories to be maintained. None of this happens overnight so always aware that any other claim of weight loss is just a matter of losing water weight and some muscle, none of which will help you in the long run.
Calories Matter But What You Eat Matters More For Weight Loss

You might think that since weight gain depends on calories from any source that you can eat pretty much anything as long as your intake remains within the boundaries of what you need to maintain an energy balance. This is the premise behind many diet fads and food manufacturers love it as it advocates that you can eat what you want- junk food included. As attractive as this may sound to the consumer however the logic behind it is severely flawed. Firstly, the energy balance principles derived from the laws of thermodynamics don’t account for the fact that we are human beings and that many other factors come into play where weight gain is concerned. If you follow the idea of weight gain being a matter solely based on energy balance then there should come a time when weight gain stops as the increased mass and consequent increase in metabolism compensates for the extra calories being consumed.[2] The larger you get the more calories you burn and yet in the real world this almost never happens as people continue to gain weight throughout the course of their lives.[3] Our behavior and eating patterns play a major part in weight gain and food manufacturers are quite aware of this. If you only ate junk food in amounts meeting your body’s energy requirements it might work, but those foods are engineered by folks who spend a lot of time, money and effort studying and mastering the creation of foods that are designed to make you eat more. Foods containing multiple ingredients that can be almost addictive. Studies have found that ingredients in many processed foods such as high fructose corn syrup make us eat and drink far more than we should and thus contributing to the continued weight gain.[3] The same applies to high fat foods and high sugar foods which activate our reward system in a way that makes us eat more and more- past what our body requires for energy balance.[5] Combine that with the ease of consumption of liquid calories in sodas and juices and you have a situation where it is very difficult for moderation to occur. More importantly, by continued consumption of junk foods you simply can’t learn the healthy eating habits needed to keep weight off. Nor can you learn how to select single ingredient natural foods which are much easier to eat in moderation than their distant multi-ingredient, processed cousins. Long term weight loss comes from a change in lifestyle and thus anything that keeps you away from doing so makes it far less likely that you’ll be successful in your long-term weight loss endeavors. (See my article on Controlling Your Eating Habits- How To Deal With Cravings.)
So, what about the stories of those who lost significant amounts of weight in a short time from low carbohydrate diets? Again, an understanding of the science behind how our body works gives us some insight. Carbohydrates are stored in our body as glycogen, which is a combination of glucose and water. When you reduce your carbohydrate intake your glycogen levels will drop, and you will lose some of that water in the process. That’s the weight you’ll see lost on the scale, but it is all water weight, and does nothing to reduce your fat stores. An effect that is at best temporary and a distraction from the practice of watching your portions and eating in a consistently better fashion with a diet that includes all food groups in sensible quantities. There aren’t any shortcuts and faced with the abundance of so many energy dense foods and our inability to accurately estimate how many calories we consume [4] it is a difficult task indeed. A slight excess in caloric intake can go a long way as eating only 40 Calories more than our daily energy requirements can add an extra 40 lbs of fat over the course of ten years. It’s a sobering thought as we are talking about eating just about half an apple more than your body needs on a daily basis and given the superabundant food supply we have today, and how easy it is to eat more than you should. it becomes a bit easier to understand one of the key reasons why most of us are overweight and our current obesity crisis. It also highlights how easy it is to regain weight and why short term solutions that involve extreme deprivations and excessive amounts of activity should always be avoided as it leads to overconsumption in the future. An understanding of how calories work make us aware that we have to find a steady approach that you can stick to for years. Not weeks or months, but long term and that rules out just about all fads that come and go. It might be an uphill battle, but it’s one that can be won with a consistent approach that emphasizes lifestyle change and not immediate weight loss results.
High Intensity Bodyweight Training: Ballistic Pushups & Dips!
This was a tough one!
Starts out with ballistic push ups (like clap pushups but without the clap as my wrist is still not 100%) nonstop for 20 reps, then all out on dips for 10 reps.
To say it was painful would be an understatement, but you just have to push through and keep on going.
Still training, hope you are too and as always, Excelsior!!! #naturallyintense
#hometraining #homeworkout #homeworkout #highintensitytraining #naturalbodybuilder #naturalbodybuilding #fitover40 #naturalbodybuildingvideos #chestday #chesttraining #naturalbodybuildingtips #pushups #dips #bodyweighttraining #highintensitytrainingtips #drugfreebodybuilding #calesthenics
Kevin's Unconventional Biceps Training- 3-6 Minutes a Week!
In this video I go over my biceps training using the Naturally Intense High Intensity Training protocols that helped me go from having arms measuring 11.5 to 12 inches to 18 inches drug free!
It's an unconventional approach for certain, but it's one that's helped my arms grow and the hundreds of men and women I have trained over the past 30 plus years.
Now, my success isn't due to being genetically gifted, as it took me the better part of 11 years to get my arms up to those measurements.
Which is significant as it works and been been proven time and time again to work for the average man or woman trying to grow their arms without drugs.
It's my hope that these high intensity training protocols can help you as much as they helped me!
Click on my bio link to see the full video on my YouTube channel and thanks as always for taking the time to look at my work!!! Excelsior!!! #naturallyintense
#highintensitytraining #naturalbodybuilder #naturalbodybuilding #fitover40 #naturalbodybuildingvideos #armworkout #bicepsworkout #naturalbodybuildingtips #biceps #armtraining #highintensitytrainingtips #drugfreebodybuilding #barbellcurls
At the Lancaster Classic Day 2 Elimination Rounds Against European Champion, and World Record Holder Leo Pettersen @leo_barebow_archer
I don't talk much about it but I'm also a competitive barebow archer (surprise!) and last Saturday I had the honor of making it to Day 2 at the Lancaster Archery Classic in the Barebow Division, as I made the top 64 out of 267 competitors and had a chance to shoot with some of the greatest barebow shooters on the planet!
I didn't make it past Leo, but it was a real rush to be there and a huge thanks to my coach, Joe MyGlyn @prolinearchery for helping me get there.
Thanks as well to my good friend @sean_chan33 for all of his help from the very start, to my line buddy Aaron Shea for taking the shot and showing up to support!
My thanks as well to rob_kaufhold for putting on and promoting one of the best archery tournaments on earth!
Thanks also to to everyone who took the time to send a supporting word and I am looking forward to next year!!! #naturallyintense #barebow
#lancasterclassic #lancasterarcheryclassic2024 #lancasterarchery #archery #fitover40 #barebowrecurve #targetarchery
Dumbo, Brooklyn circa 2004
This shot was taken as part of the promotion for my Naturally Intense DVD and was about a year after my last bodybuilding competition.
It was a grueling photoshoot.
We started at about 10 am and finished around 4pm and I was completely spent, but the more we shot the sharper I looked, so we kept on going.
It's nice to look back from time to time and as tired as I was, we all had a blast!
My thanks to @stephanie_corne_artwork, @https://pulse.ly/itgnag2dec and @ftaz1 for taking the shots!!!
Thanks for watching and as always, Excelsior!!! #naturallyintense
#naturalbodybuilder #naturalbodybuilding #throwback #fifthavenuegym #5thavenuegym #drugfreebodybuilding #naturalbodybuildinglifestyle #gymlife #gymmotivation #naturalbodybuildingmotivation #bodybuilding #blackandwhite #instablackandwhite #bnw
Can You Build An Impressive Physique Training Only At Home?
Absolutely!
I stopped training in commercial gyms as of March 2020 and have been training at home ever since.
Initially I was admittedly worried that I might lose some of my gains or not make as much progress, but that certainly wasn't the case.
I've consistently continued to improve with my high intensity workouts and muscles have no idea where they are training.
As long as the criteria of adequate intensity and overload are met, there will be an adaptive response and your muscles will get bigger and stronger.
So don't worry at all about where you train, focus instead of what will be the best way for you to always be training!
Thanks for watching and as always, Excelsior!!! #naturallyintense
Kevin's Three Day Training Spilt!
For the past 33 years I have trained three times a week with Naturally Intense High Intensity Training workouts lasting 10, 15 to 20 minutes max.
It's a training split tried and testes not only in it's helping me realize my goal of becoming a successful natural bodybuilder, but it's also helped hundreds of men and women over the past three decades.
I have tested just about every possible training split imaginable and for this particular style of high intensity training, this particular grouping consistently yields fantastic results.
I hope it helps you as much as it's helped me over the years and thanks so much for taking the time to look at my work.
Keep training hard and Excelsior!!! #naturallyintense
Excelsior!!! #naturallyintense
#trainingsplit #3daytrainingsplit #threedaytrainingsplit #naturalbodybuilding #naturalbodybuilder #naturalbodybuildingvideo #naturalbodybuildingmotivation #naturalbodybuildingtips #drugfreebodybuilding #bodybuilding #highintensitytraining #highintensitytrainingtips
405 Stiff Leg Deadlift for 7 Reps! High Intensity Training.
First leg workout of the year and already pushing it!
I haven't done a stiff leg deadlift over 315lbs for about 3 years at this point, and I did my last set with 315lbs and comfortably got to 10 reps and decided I had far too much gas left in the tank and that I should go up in weight.
So I did.
I figured I might get a solid 6 reps in, but I made it to 7 and I think I could have gone on to get a full 10 reps BUT that's when good judgement prevailed.
As a bodybuilder having not trained this heavy for so many years, the shock of this much weight would be more than enough to stimulate muscle growth, and doing more reps wouldn't yield any greater returns, only increase the likelihood of injury.
It's not about the numbers, it's about training to a point where you achieve your goal, and it's important to have a goal in mind as a bodybuilder based on increasing muscle mass rather than hitting a particular number.
Besides, if in my 20's I never did more than 405lbs on a stiff leg deadlift, it doesn't make any sense going heavier than when I am almost 50!
Could I deadlift more at this point?
Absolutely but just because you can doesn't mean you should!
So keep those weights in a good working range, keep it safe and as always Excelsior!!! #naturallyintense
#hometraining #homeworkout #homeworkout #roguerack #highintensitytraining #naturalbodybuilder #naturalbodybuilding #fitover40 #naturalbodybuildingvideos #backworkout #naturalbodybuildingtips #backtraining #highintensitytrainingtips #drugfreebodybuilding #fitoverforty #deadlift
Turning 50 in a few months...
Not much of a big deal for me as I still feel pretty much the same but I hope that my example helps show what can be done with a lifetime commitment to eating well and training consistently!
Thanks for coming along on the journey and as always, Excelsior!!! #naturallyintense
#naturalbodybuilder #naturalbodybuilding #healthylifestyle #fitover40 #drugfreebodybuilding #naturalbodybuildingmotivation #natty #fitness
Please note that all material is copyrighted and DMCA Protected and can be reprinted only with the expressed authorization of the author.
Related Articles:
Food And Self Control: How To Reduce Cravings
Understanding Glycemic Index and Weight Loss
Click To Get A Copy Of Kevin’s Free Ebook On The Role Of Exercise In Reducing Abdominal Fat!
Featured everywhere from the Wall Street Journal to network TV, Kevin Richardson is the international fitness consultant for UNICEF, natural bodybuilding champion, creator of Naturally Intense High Intensity Training and one of the top personal trainers in New York City.
References For Understanding How Calories Relate To Weight Loss
1. National Health & Nutrition Examination Survey- Intake of Calories and Selected Nutrients for the United States Population, 1999-2000
2. Kushner, Robert. Treatment of the Obese Patient (Contemporary Endocrinology). Totowa, NJ: Humana Press. 2007
3. Wells JC, Siervo M. Obesity and energy balance: is the tail wagging the dog? Eur J Clin Nutr. 2011
4. Kopelman PG, Caterson IG, Stock MJ, Dietz WH. Clinical obesity in adults and children: In Adults and Children. Blackwell Publishing 2005
5. Erlanson-Albertsson C. How palatable food disrupts appetite regulation. Basic & Clinical Pharmacology & Toxicology 2005
6. Krzywicki HJ, Consolazio CF, Matoush LO et al. Am J Clin Nutr 1968
7. Drenick EJ. The effects of acute and prolonged fasting and refeeding on water, electrolyte, and acid-base metabolism. In: Maxwell MH, Kleeman CR, eds. Clinical Disorders of
Fluid and Electrolyte Metabolism. New York: McGraw-Hill, 1980







